NCERT Solutions for Class 9 Science Chapter 1 Matter in Our Surroundings
NCERT Solutions for Class 9 Science (chemistry) Chapter 1 Matter in Our Surroundings are given below. In these solutions, we have answered all the intext and exercise questions provided in NCERT class 9 science textbook. Class 9 NCERT Solutions Science Chapter 1 provided in this article are strictly based on the CBSE syllabus and curriculum. Students can easily download these solutions in PDF format for free from our app.
Class 9 Science Chapter 1 Textbook Questions and Answers
Intext Questions
Page No 3
Question 1: Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold drink, smell of perfume.
Answer: Chair, air, almonds, and cold-drink are matters.
Explanation: Things that occupy space and have some mass are called matter. Since chair, air, almonds and cold-drink occupy some space and have some mass, so these are matter.
Question 2: Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer: The smell of hot sizzling food reaches severed meters away, as the particles of hot food have more kinetic energy and hence the rate of diffusion is more than the particles of cold food.
Or
Smell of anything comes because of gases emanating from the given thing. The smell reaches to us because of diffusion of gas. The rate of diffusion increases with increase in temperature. This happens because of higher kinetic energy due to higher temperature. That is why smell of hot sizzling food reaches to us from several feet. On the other hand, the kinetic energy of gases emanating from cold food is low because of lower temperature. Due to this, we need to move closer to a cold food to take its smell.
Question 3: A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer: A diver is able to cut through water in a swimming pool. This shows that the particles of water have intermolecular space and has less force of attraction.
Question 4: What are the characteristics of particles of matter?
Answer: The characteristics of particles of matter are:
- Particles of matter have spaces between them.
- Particles of matter are continuously moving.
- Particles of mater attract each other.
Page No. 6
Question 1: The mass per unit volume of a substance is called density (density = mass/volume). Arrange the following in order of increasing density − air, exhaust from chimney, honey, water, chalk, cotton, and iron.
Answer: The given substances in the increasing order of their densities can be represented as:
Air < Exhaust from chimney < Cotton < Water < Honey < Chalk < Iron
Explanation: Air is the mixture of gases. Chimney exhaust is also a mixture of gases; along with some heavier particles, such as ash. This makes the density of chimney exhaust more than air. Cotton is a porous solid and which has lot of air trapped within pores. This makes its volume more than water. Therefore, it is less dense than water.
Question 2: (a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy and density.
Answer: (a) The differences in the characteristics of states of matter are given in the following table.
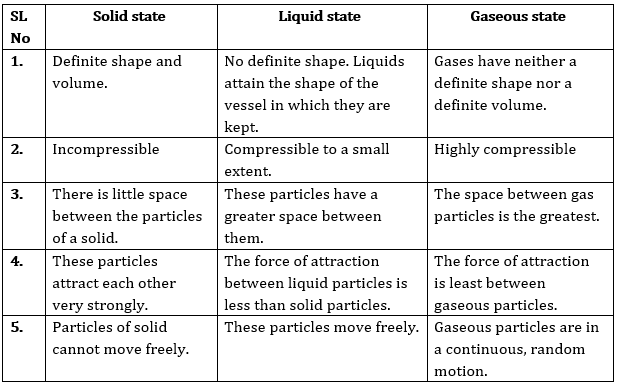
Or
The difference in the characteristics of the three states of matter.
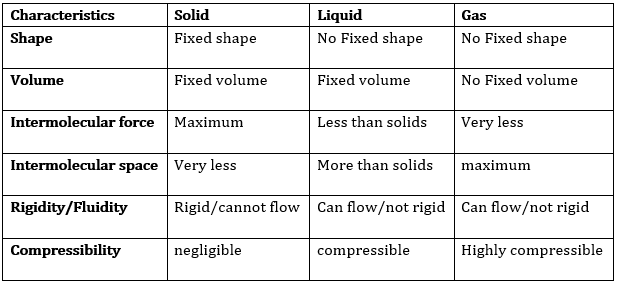
(b) Rigidity: The greatest force of attraction between particles and close packing of particles make solids rigid. Rigidity is one of the unique properties of solids. Because of rigidity, a solid can resist from getting distorted. Because of rigidity a solid has definite shape and volume. Rigidity is negligible in fluid and gas.
Compressibility: Compressibility is one of the most important characteristics of gas. Because of lot of space between particles, a gas can be compressed to a great extent.
Liquid and solid cannot be compressed because of the least space between their particles.
Fluidity: The ability to flow is called fluidity. The less force of attraction and more space between particles make liquid and gas to flow. That’s why liquid and gas are called fluid.
Filling of a gas container: Liquids do not fill a gas container completely, while gases fill the gas container completely in which it is kept. This is because the particles of gas can move in all the directions.
Shape: Solids have fixed shape. Liquid and gas take the shape of the container in which they are kept. This happens because of less force of attraction and more kinetic energy between particles of liquids and negligible force of attraction and highest kinetic energy between particles of gas.
Kinetic energy: The kinetic energy of particles of solid is the minimum. They only vibrate at their fixed position. The kinetic energy of particles of liquid is more than that of solid. But they can slide above one another. The kinetic energy of particles of gas is the maximum.
Density: The mass per unit volume of a substance is called density. The density of solid is highest, of liquid is less than solid and of gas is minimum.
Question 3: Give reasons:
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Answer: (a) There is little attraction between particles of gas. Thus, gas particles move freely in all directions. Therefore, gas completely fills the vessel in which it is kept.
(b) Because of negligible force of attraction between particles of gas, the particles of gas have the highest kinetic energy. These properties enable the particles of gas to move in all directions and hit the walls of container from all sides. Because of this a gas exerts pressure on the walls of the container in which it is kept.
(c) A wooden table has a definite shape and volume. It is very rigid and cannot be compressed i.e., it has the characteristics of a solid. Hence, a wooden table should be called a solid.
(d) Particles of air have large spaces between them. On the other hand, wood has little space between its particles. Also, it is rigid. For this reason, we can easily move our hands in air, but to do the same through a solid block of wood, we need a karate expert.
Or
Since, air is gas, so its particles are loosely packed and there is negligible force of attraction between its particles. Because of that we can easily move our hand in air. But wood is a solid, so the force of attraction between its particles is greatest. The particles of wooden block are closely packed. That’s why we cannot move our hand through a solid block of wood. However, a karate expert can exert required pressure to break the great force of attraction of the particles of a solid wooden block.
Question 4: Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer: During freezing of water, some space between the particles of water is left vacant with some air trapped between them. These empty spaces having air in them makes the density of ice; lower than that of water. That’s why ice floats on water.
Page No. 9
Question 1: Convert the following temperatures into the Celsius scale.
(a) 300 K
(b) 573 K
Answer: (a) 300 K = (300 − 273)°C = 27°C
(b) 573 K = (573 − 273)°C = 300°C
Question 2: What is the physical state of water at
(a) 250°C
(b) 100°C
Answer: (a) Water at 250°C exists in gaseous state.
(b) At 100°C, water can exist in both liquid and gaseous form. At this temperature, after getting the heat equal to the latent heat of vaporization, water starts changing from liquid state to gaseous state.
Question 3: For any substance, why does the temperature remain constant during the change of state?
Answer: During a change of state, the temperature remains constant. This is because all the heat supplied to increase the temperature is utilized (as latent heat) in changing the state by overcoming the forces of attraction between the particles. Therefore, this heat does not contribute in increasing the temperature of the substance.
Question 4: Suggest a method to liquefy atmospheric gases.
Answer: Atmospheric gas is liquefied by increasing pressure and decreasing temperature.
PAGE NO. 10
Question 1: Why does a desert cooler cool better on a hot dry day?
Answer: Desert cooler works on the basis of evaporation. In hot and dry days the moisture level is very low in atmosphere which increases the rate of evaporation. Because of faster evaporation, cooler works well. That’s why desert cooler cool better on a hot dry day.
Or
When a liquid evaporates, the particles of the liquid absorb energy from the surroundings to compensate the loss of energy during evaporation. This makes the surroundings cool.
In a desert cooler, the water inside it is made to evaporate. This leads to absorption of energy from the surroundings, thereby cooling the surroundings. Again, we know that evaporation depends on the amount of water vapour present in air (humidity). If the amount of water vapour present in air is less, then evaporation is more. On a hot dry day, the amount of water vapour present in air is less. Thus, water present inside the desert cooler evaporates more, thereby cooling the surroundings more. That is why a desert cooler cools better on a hot dry day.
Question 2: How does water kept in an earthen pot (matka) become cool during summers?
Answer: Water from porous wall of earthen pot evaporates continuously, which lowers the temperature of water kept in the earthen pot. In summer moisture level is very low in the atmosphere, which increases the rate of evaporation as evaporation is inversely proportional to the moisture level in atmosphere. That is why in summer water kept in earthen pot becomes cool.
Question 3: Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer: When we put some acetone or petrol or perfume on our palm, it evaporates. During evaporation, particles of the liquid absorb energy from the surrounding or the surface of the palm to compensate for the loss of energy, making the surroundings cool. Hence, our palm feels cold when we put some acetone or petrol or perfume on it.
Question 4: Why are we able to sip hot tea or milk faster from a saucer than a cup?
Answer: When hot tea or milk is kept in a saucer, the liquid is exposed over a larger surface area as compared to in case of the liquid being kept in a cup. The larger surface area enables the faster cooling. That’s why we are able to sip hot tea or milk faster from a saucer rather than from a cup.
Question 5: What type of clothes should we wear in summers?
Answer: In summer, it is preferred to wear light-coloured cotton clothes because light colour reflects heat and cotton materials have pores that absorb sweat, facilitating their evaporation hence causing a cooling effect in the skin.
Exercises
Question 1: Convert the following temperatures into the Celsius scale.
(a) 293 K
(b) 470 K
Answer: Temperature in Celsius scale = Temperature in Kelvin scale – 273
(a) 293K= (293 – 273)°C = 20°C
(b) 470K= (470 – 273)°C = 197°C
Question 2: Convert the following temperatures into the Kelvin scale.
(a) 25°C
(b) 373°C
Answer: Temperature in Kelvin scale = Temperature in Celsius scale + 273
(a) 25°C = (25+273)K = 298K
(b) 373°C = (373+273)K = 646K
Question 3: Give reasons for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
(b) We can get the smell of perfume while sitting several metres away.
Answer: (a) At room temperature, naphthalene balls undergo sublimation wherein they directly get converted from a solid to a gaseous state without having to undergo the intermediate state, i.e., the liquid state.
(b) Perfumes vaporize very fast and its vapours diffuse into air easily. That is why we can smell perfume sitting several meters away.
Question 4: Arrange the following in increasing order of forces of attraction between the particles – water, sugar, oxygen.
Answer: Oxygen < Water < Sugar.
Explanation: Oxygen is a gas, thus force of attraction is negligible between particles. Water is a liquid, thus force of attraction between particles is more than liquid and less than solid. Sugar is a solid, thus force of attraction between particles is greatest.
Question 5: What is the physical state of water at —
(a) 25°C
(b) 0°C
(c) 100°C?
Answer: (a) At 25°C – water is in liquid state.
(b) At 0°C – water is in solid state.
(c) At 100°C – water is in transition state, i.e. in liquid and gas both.
Question 6: Give two reasons to justify:
(a) water at room temperature is a liquid.
(b) an iron almirah is a solid at room temperature.
Answer: (a) At room temperature (25 °C), water is a liquid because it has the following characteristic of liquid:
(i) Water has definite volume, but not definite shape as it takes the shape of the container in which it is kept.
(ii) Water flows at room temperature.
(b) An iron almirah is a solid at room temperature because:
(i) It has definite shape.
(ii) It has definite volume.
Question 7: Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer: At 273K ice requires more latent heat to melt into water, while water at 273K requires less latent heat; to come to the room temperature. So, ice at 273 K is more effective in cooling than water at the same temperature.
Question 8: What produces more severe burns, boiling water or steam?
Answer: Steam produces more severe burns than boiling water. This is because steam has more energy than boiling water, present in it in the form of latent heat of vaporization.
Question 9: Name A, B, C, D, E and F in the following diagram showing change in its state:
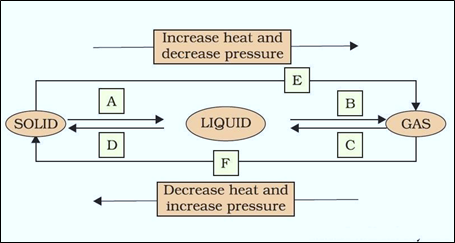
Answer:
A: Melting (or) fusion (or) liquefaction
B: Evaporation (or) vaporization
C: Condensation
D: Solidification
E: Sublimation
F: Sublimation
Class 9 Science NCERT Solutions Chapter 1 Matter in Our Surroundings
CBSE Class 9 Science NCERT Solutions Chapter 1 helps students to clear their doubts and to score good marks in the board exam. All the questions are solved by experts with a detailed explanation that will help students complete their assignments & homework. Having a good grasp over CBSE NCERT Solutions for Class 9 Science will further help the students in their preparation for board exams and other competitive exams such as NTSE, Olympiad, etc.
NCERT Solutions for Class 9 Science Chapter 1 PDF
Below we have listed the topics discussed in NCERT Solutions for Class 9 Science Chapter 1. The list gives you a quick look at the different topics and subtopics of this chapter.
| Section in NCERT Book | Topics Discussed |
|---|---|
| 1.1 | Physical Nature of Matter |
| 1.2 | Characteristics of Particles of Matter |
| 1.3 | States of Matter |
| 1.4 | Can Matter Change its Shape? |
| 1.5 | Evaporation |